Repeatedly in Holland Crop Circles
by
Nancy Talbott
BLT Research Team Inc.
www.bltresearch.com
As has occurred repeatedly in crop circles found in the fields around Hoeven (and also in conjunction with other anomalous incidents which occur at the home of Robbert v/d Broeke), a very fine-grained white powder appeared again on October 5, 2009 -- this time in the center of a small new circle which had been added to a series of rings discovered previously on September 27th.
On September 6th, a month after I had returned to the States after being with Robbert in 2009 and watching him as he took dozens and dozens of anomalous photos in crop circle fields using many different people's cameras (http://www.bltresearch.com/robbert/ufophotos.php), a small ring with three "squashed" areas around the outside of the ring was found in the grass in this same field where the white powder appeared a month later. As is often the case, Robbert had "seen" this ring appearing as an image in his "mind's eye" during the night.
On the night following it's discovery Robbert and his friends Ellen Gomis and Roy Boschman went to the field to take photographs, since they know that very often Robbert will obtain anomalous images in crop circle fields shortly after new formations appear. At first the photos were all perfectly normal. But then Roy handed his camera to Robbert and a few anomalies began to appear, followed by multiple "light-ball UFO" images which occurred when Robbert aimed the camera at himself.
Three weeks later -- during the night of September 27 -- another series of thin rings appeared, spread out across a large area of the field. And, again, Robbert could "feel" the new rings were occurring. Because Robbert no longer goes out into the fields at night by himself he called Roy and also Peter Vanlaerhoven of the Dutch crop circle association (DCCA), so they could document the new rings before the grasses recovered (which they often do fairly quickly).
For additonal photos of these various ring formations see the DCCA web-site (http://www.dcca.nl/2009/2009-uk.htm).
A week after these photos were taken Robbert again, while at home with his family on the night of October 5th, "saw" a small new circle appearing in the field with something "bright white" in it's center and again notified both Roy and the DCCA group immediately.
Because previous lab analyses (by BLT's analytical chemist Phyllis Budinger and materials scientist Dr. Jaap J. Boon of the FOM Institute in Amsterdam) of similar-looking white powders found in association with other events around Robbert had repeatedly revealed that the material was magnesium carbonate (see links provided at the beginning of this report), in a chemical formulation that is used as a fire-proofing agent or as a fire-retardant, members of the DCCA group decided to test for themselves to see whether the material would burn.
|
Mass Spectroscopy Analysis

![]()
17100 Wood Acre Trail
Chagrin Falls, Ohio 44023
Phone/Fax: 440-708-2211
Email: BudingerPA@MSN.com
TECHNICAL SERVICE RESPONSE NO.: UT071
Subject: |
Addendum to UT069: Identification of a White Powder Found in a Crop Formation (Hoeven, Holland, October 5, 2009) |
| Date: | June 22, 2010 |
| Requested By: | Nancy Talbott, BLT Research |
| Reported By: | P. A. Budinger Analytical Scientist |
|
Background/Objective:
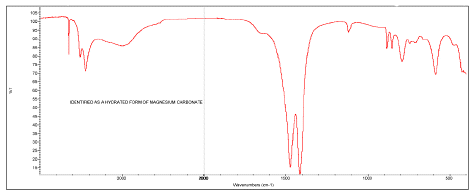
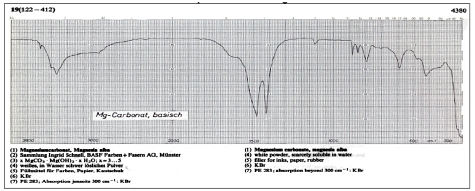
A white powder found in a crop formation in Holland (October 5, 2009) was identified by this laboratory as a hydrated form of magnesium carbonate, specifically with formula (MgCO3)4
·Mg(OH)2
·5H2O. In the analytical report (Technical Service Report: UT069) a recommendation was made to do additional ICP-MS analysis to determine both the magnesium isotopic ratios1 and purity. More of the sample was submitted on February 24, 2010 from this event to do the recommended analysis.
·The white powder is confirmed by FT-IR2 to be the same hydrated form of magnesium carbonate (approximate formula of (MgCO3)4
·Mg(OH)2
·5H2O) as the previous sample received October 5, 2009. ·ICP/MS shows the isotopic ratios of this magnesium are normal for terrestrial magnesium. ·The ICP elemental results show the magnesium carbonate is 99.99% pure. This surprising result would suggest that the material is chemically made and/or refined from a natural source. Clearly, at this purity it is beyond agricultural use, which appears to be about 85% purity (http://www.sangamproducts.com/Magnesium.html). Magnesium carbonate at this 99.99% purity could be found in a laboratory storeroom. Alfa Aesar sells 99.996% pure magnesium carbonate at $717.00 per 100 grams (http://www.alfa.com/en/GP100w.pgm?DSSTK=010796). This is rather expensive, and copious amounts of this material have been recovered from various formations and other bizarre events related to Robbert v/d Broeke.3 ·What is also puzzling is that material recovered from an open field should show evidence of contamination by airborne dust particles. Of course one could argue that the dust would only be on the surface so, if the samples were taken from the center of the pile, one might avoid contamination. But if one just shoveled the material into a bucket while sampling, as was the case here, one would expect mixing. Procedure:
More white powder from the October 5, 2009 crop formation in Holland was submitted on February 24, 2010. An infrared spectrum was obtained of the new sampling to confirm it was the same material as submitted on October 5, 2009. The spectrum was obtained of the sample on the Thermo Electron Avatar 360 spectrometer using the Smart Herrick diamond sampling accessory.
|
|
BLT's consulting analytical chemist has been fairly certain that the purity level of the many samples of white powder she has worked on related to events involving Robbert was highly refined, but FT-IR testing alone cannot establish this fact conclusively. We therefore decided to obtain the ICP-MS analysis in addition to FT-IR this time -- because it does establish the presence of elements other than those normally found in magnesium carbonate, determining purity of the sample down to the parts-per-million level.
So how does one approach a reasonable interpretation of these facts? Neither Robbert nor any of his friends are financially able to afford this material and none of them are aware of a professional laboratory supply company anywhere in the area. If there is someone in the area wealthy enough to afford this material, how could they know (in multiple incidents now, for more than ten years) exactly when to arrive and at which field? How did they manage to avoid being seen in 2007 on the night when Robbert and most of his family and Mr. and Mrs. Gomis were present in the field at "Woodenhead" as a new crop circle actually formed with the white powder in its center?
convincing those who do not know it...but for the sake of defending those that do." --William Blake
|